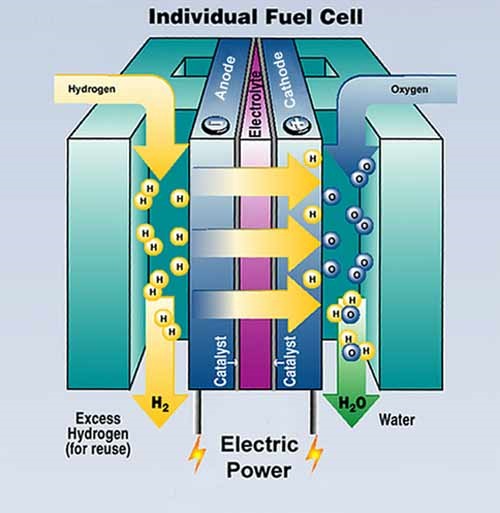 
Hydrogen enters the fuel cell via the anode.Composed of two electrodes (an anode and a cathode) separated by an electrolyte membrane, a typical hydrogen fuel cell works in the following way: Similar to batteries, a fuel cell is a device that converts energy stored in molecules into electricity through an electrochemical reaction. Tomorrow, it could potentially power everything from low-carbon cities and regions to portable computing devices to future zero-emission aircraft. power a variety of transportation modes such as cars, buses, trains and forklifts.replace grid electricity for critical-load facilities like data centres.provide emergency backup power to critical facilities like hospitals.Today, hydrogen fuel cell technology is being used for a variety of applications, including to: As the story goes, then-US President Richard Nixon famously said: "Without you Tom, we wouldn't have gotten to the moon.” It remained a scientific curiosity until the 20th century, when English engineer Francis Thomas Bacon matured the original idea to develop the world’s very first hydrogen-oxygen fuel cell in 1932.īacon’s fuel cell was such a success that it has been used by the space industry to power satellites and rockets for space exploration programmes, including Apollo 11, since the 1960s. At the time, he called his invention a “gas voltaic battery.” Unfortunately, it did not produce enough electricity to be of much use. In 1838, judge-turned-scientist Sir William Grove came up with a novel idea: to construct a cell consisting of two separate sealed compartments, each of which was fed by either hydrogen or oxygen gas. In a strategic partnership with automotive systems supplier ElringKlinger, Airbus is investing to mature fuel cell propulsion systems for the aviation market. It would be more beneficial in a power plant.H ydrogen fuel cells are emerging as a high-potential technology that offers significant energy efficiency and decarbonisation benefits to a range of industries-including automotive and heavy transport. For example, an MCFC is inappropriate for use in the home as it has a very high operating temperature of up to 650 ☌. These different kinds of fuel cells can be used depending on the appliance and how much electricity is required. A proton exchange membrane (PEM) can output between 50 and 250 kWs of electricity and a molten carbonate fuel cell (MCFC) can output a huge two megawatts of electricity. It’s thought that their efficiency is around 70 per cent and they can output anything from 300 watts to five kilowatts (kW) of electricity. These run using compressed hydrogen and oxygen. One of the most common types of fuel cell is an alkali fuel cell. A fuel cell will produce just under one volt of electricity and so hundreds or even thousands need to be stacked to run a car. One hydrogen fuel cell hardly produces any electricity - this is why they need to be stacked together so that the electricity output is usable. How much electricity does a hydrogen fuel cell produce? 
Hydrogen costs around £12 per kg, therefore a tank capacity of around five kgs could cost between £40 and £60 to fill. This range is much greater than most electric cars on the market. You could drive for 500 km before needing to refill, making a hydrogen fuel cell car very similar in cost and range to a petrol vehicle. To put this in perspective, around one kg of hydrogen equals nearly five litres of petrol. In a typical fuel cell vehicle, you can expect to use around 0.8 kilograms (kg) of hydrogen per 100 kilometres (km). A further bonus is that any excess electricity can be sold back to the grid. For example, in a CHP boiler system, the heat that is created can be used to further warm the water in the system for very high energy efficiency. Not only is the electricity being put to use, but the heat byproduct can be applied elsewhere too. If the heat that is produced by fuel cells in a combined heat and power (CHP) system is reused, the efficiency increases even more. This depends on how the fuel cell is being used and is usually unachievable in real world applications. The department goes on to state that the maximum efficiency that a combustion engine could achieve is around 58 per cent, however fuel cells have a theoretical maximum efficiency of 85 to 90 per cent. This number could vary depending on the size and weight of the car, as well as the kinds of roads being driven on. But how efficient are hydrogen fuel cells, and how much hydrogen needs to be inputted in order to get a usable electricity output?Īccording to the US Department of Energy Hydrogen Program, a standard fuel car with a combustion engine runs at around 20 per cent efficiency, whereas vehicles that run using hydrogen fuel cells are around 40 to 60 per cent efficient.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
